Proton charge and electron charge9/5/2023  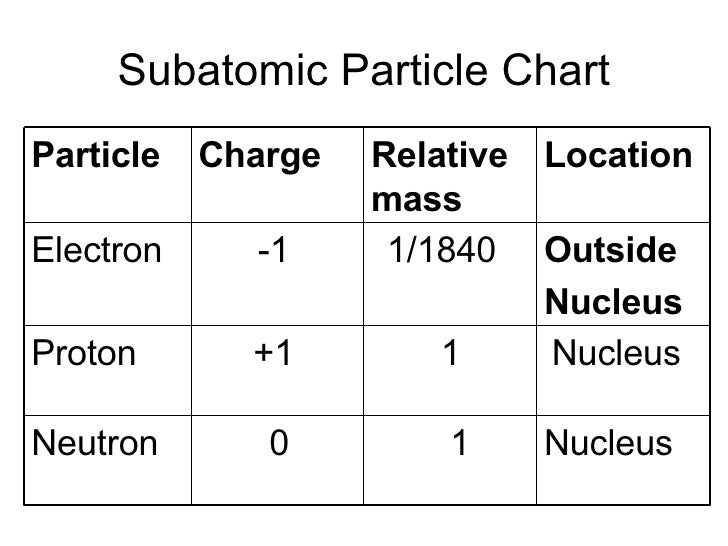 
He accepted hydrogen as an elementary particle by 1920 and named it a proton. The ejection of hydrogen nuclei from nitrogen was shown by Ernest Rutherford in 1919 on the bombardment of alpha particles. Thomson identified a positive particle with a mass equal to a hydrogen atom. While observing streams of ionized molecules and atoms in gaseous form from which electrons were stripped, Wilhelm Wien and J.J. 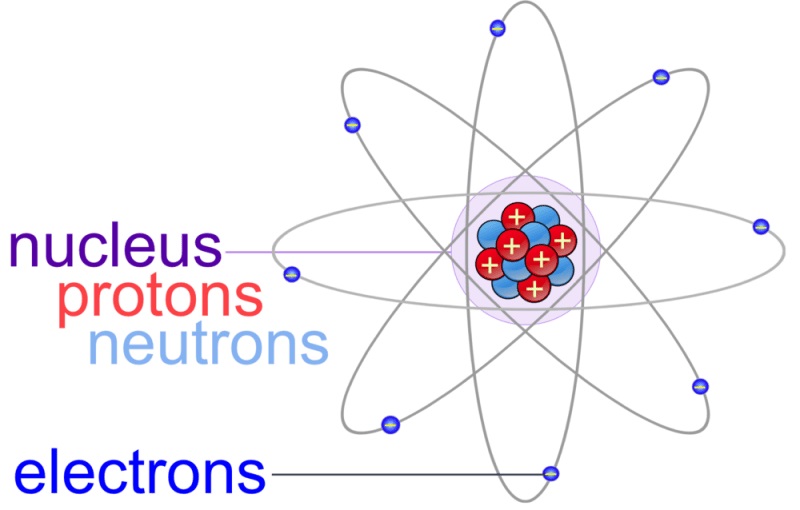
The discovery of protons was made when the earliest investigations of atomic structures were back in history. When the proton count in a nucleus is equal to the electron count orbiting around it, the atom is supposed to be electrically neutral. The same number of protons are present in every nucleus of a given element, and it defines its atomic number and position in the periodic table. Protons, along with neutrons, form all the atomic nuclei except hydrogen nuclei that have a single proton. If you are wondering what is a proton, the proton definition is that it is a subatomic entity having a positive charge that is equivalent in magnitude to an electron charge unit and a rest mass equal to 1.67263 × 10 -27 kg. This energy actually came from the lost mass, according to Einstein’s formula E = mc 2 What is a Proton? The energy released in this process fuels most types of stars in both their red giant and normal phases. The method by which carbon is formed is by nuclear fusion of hydrogen into helium and helium to carbon. These atoms can further be divided into protons, electrons, and neutrons.Ī carbon atom comprises six neutrons and six protons, which is approximately 0.8% lighter than the individual particles that build it. Cells can be broken down into organelles organelles can be divided into individual molecules, which can be further broken down into atoms. If those are broken down further into cells, those cells would still combine to give the same mass as you. If your body is broken down into individual fat, bones, and organs, those will combine to give the entire human being. If you break up the particles that form the human body into smaller bits, you will find that at every step of the way, the whole equals the sum of the individual parts, especially for mass. A proton’s mass is 1840 times more than that of an electron. It may be claimed that the mass of a hydrogen atom is equal to the mass of a proton because an electron’s mass is thought to be insignificant. One electron and one proton make up an atom of hydrogen. The proton mass is the same as the mass of an atom of hydrogen. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
